Young children can get COVID-19 vaccine starting Wednesday
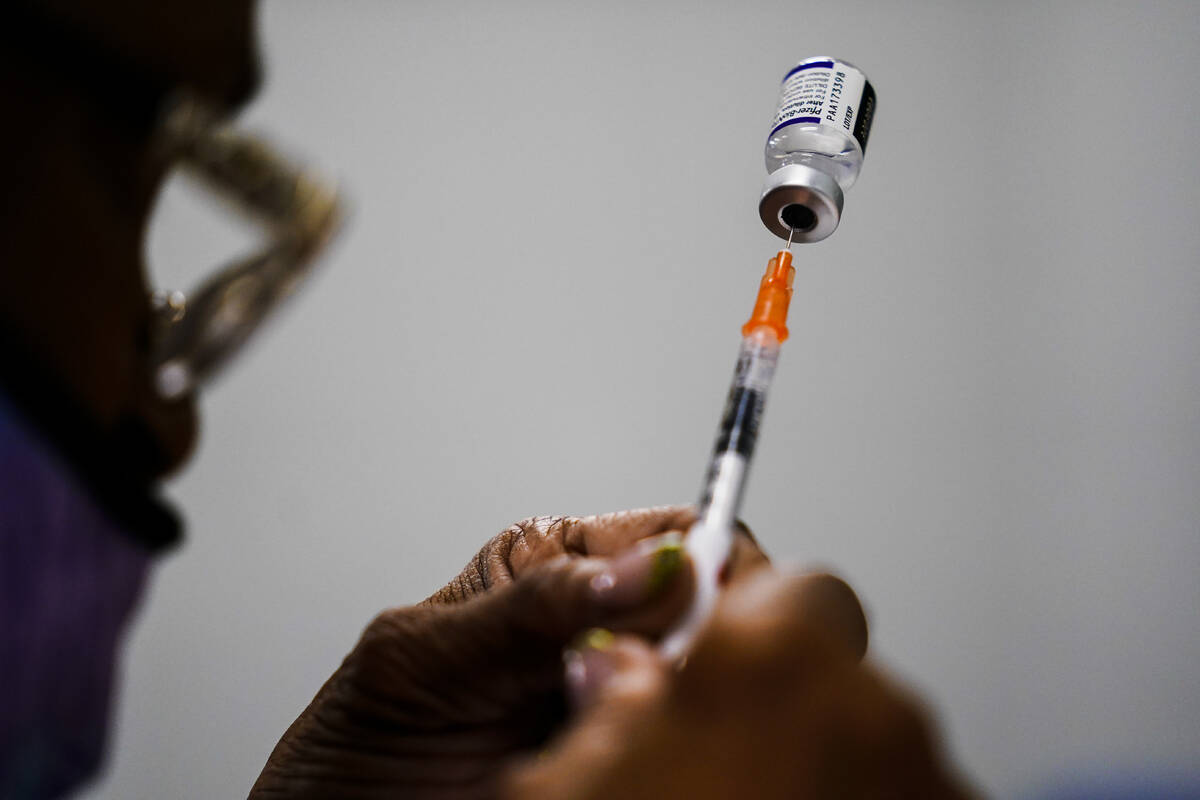
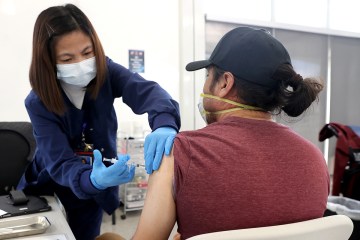
Starting Wednesday, the Southern Nevada Health District will begin offering COVID-19 vaccines to children from 6 months to 5 years of age.
The announcement came after the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices recommended both the Moderna and Pfizer vaccines and after the U.S. Food and Drug Administration gave emergency use authorization for the vaccines.
The health district will have both the Pfizer-BioNTech and Moderna vaccines available.
“We are pleased to finally be able to offer the COVID-19 vaccines to help protect our youngest residents,” Dr. Fermin Leguen, district health officer for the health district, stated in a news release. “Families have made extraordinary sacrifices these past few years to protect their children and their most vulnerable members. The availability of the vaccines for younger children is another critical step in our ongoing efforts to mitigate the impact of the pandemic on our community.”
The Pfizer vaccine is authorized for children 6 months to 4 years of age and is a three-dose series. The second dose is administered three weeks after the first dose. The third dose is administered at least eight weeks after the second dose.
The Pfizer vaccine will be available starting Wednesday at health district clinics located at the College of Southern Nevada’s West Charleston and North Las Vegas campuses, the Galleria at Sunset, the Boulevard Mall inside El Mercado, and at the Southern Nevada Community Health Center clinic at 280 S. Decatur Blvd. (located in the external structure on the south side of the building).
The Moderna vaccine is authorized for children ages 6 months to 5 years of age in a two-dose series, with the second dose administered after 28 days.
The health district will begin offering the Moderna vaccine Wednesday at the Health Center clinic at 280 S. Decatur Blvd.
On Sunday, the Western States Scientific Safety Review Workgroup completed its review of the federal process and unanimously concluded that the Pfizer and Moderna vaccines are safe and effective for children as young as 6 months old. The workgroup provided its confirmation to the governors of California, Nevada, Oregon, and Washington in the afternoon, according to a news release from Gov. Steve Sisolak’s office.
The workgroup, made up of scientists with expertise in immunization and public health, reviewed safety and efficacy data for the vaccines. It found that completion of either vaccine series produced antibody levels similar to those achieved in individuals ages 16 to 25. Vaccine reactions among infants ages 6 to 12 months and children ages 1 through 5 years old were consistent with reactions to other vaccines routinely recommended for these age groups.
The workgroup concluded that the benefits of completing either vaccine series substantially outweigh any known or likely risks. Immunization can be expected to reduce the numbers of COVID-19-related serious illnesses, hospitalizations and deaths in young children while facilitating their participation in normal educational, social and recreational activities, it said.
“Many families have been waiting for the opportunity to vaccinate their youngest members and I am happy that day has come,” Sisolak said in the news release. “Families can have the peace of mind of the thorough review process these vaccines have gone through, but I encourage families to speak to a trusted health care provider about any questions they have.”
As of June 13, the health district is reporting 13,868 cases of COVID-19 in children under the age of 4, and three deaths.
Many children who get COVID-19 will experience mild symptoms. However, some will experience more severe illness or develop long-term symptoms.
“Yes, the risk of serious illness is lower in children — at least it has been — but it’s not zero,” Dr. David Di John, a pediatric infectious disease specialist at UNLV’s Kirk Kerkorian School of Medicine, said in an interview. “And kids can get very sick from this virus.
“And so the vaccine is the best way to try to protect people from getting very sick or from dying from the disease,” added Di John, who recommends COVID-19 vaccination for all age groups.
According to the CDC, more than 591 million doses of COVID-19 vaccines have been administered in the U.S. since Dec. 14, 2020. The vaccines underwent a rigorous process to ensure their safety and effectiveness and continue to undergo the most intensive safety monitoring in U.S. history, public health authorities say.
“I would encourage parents and guardians to bring their children in to get vaccinated as soon as possible,” said Dr. Cort Lohff, chief medical officer for the health district. “This is the time of year we start reminding the community about the importance of back-to-school immunizations, and this is one more step parents can take to help ensure their children have a healthier start to the school year.”
Health district COVID-19 clinic location information is available at covid.southernnevadahealthdistrict.org.
Contact Mary Hynes at mhynes@reviewjournal.com or 702-383-0336. Follow @MaryHynes1 on Twitter. Review-Journal staffer Marvin Clemons contributed to this story.