New drug for COVID treatment remains scarce in Las Vegas
Sunrise Hospital and Medical Center, which a year ago opened one of the first centers for treating COVID-19 with monoclonal antibodies, has paused treatments while awaiting supplies of a drug shown to be more effective against the omicron variant.
The Las Vegas hospital stopped administering either the Regeneron or Eli Lilly monoclonal antibody treatments four to six weeks ago based on federal guidance that they were less effective against the omicron variant than against earlier coronavirus strains, said Dr. Steven Merta, the hospital’s chief medical officer.
Last week, the hospital received its first shipment of sotrovimab, a monoclonal antibody therapy manufactured by GlaxoSmithKline and Vir Biotechnology, shown to be more effective against omicron. But after a subsequent shipment this week, and treating fewer than 20 patients total, the hospital has run out of the drug.
“We just are anxious to get our distribution and be able to provide that to our community,” Merta said in an interview Tuesday.
Merta’s comments come following the Food and Drug Administration’s announcement Monday that it was halting use of the Regeneron and Eli Lilly treatments based on evidence of significantly reduced effectiveness against omicron. The strain now accounts for 99 percent of new COVID-19 cases in the country, according to the agency.
Most hospitals and clinics don’t have the capability to determine which virus strain a patient has, but laboratories across the country genetically analyze a sampling of positive tests.
The FDA approved GlaxoSmithKline’s sotrovimab for emergency use in May, after data indicated that the drug significantly reduced the risk of hospitalization in patients with mild to moderate COVID-19.
Treatments reduce hospitalizations
Monoclonal antibody therapies – given as a one-time, outpatient IV infusion – have been used to treat patients with mild to moderate COVID-19 who, due to age or underlying conditions, are at increased risk of becoming severely ill from the disease.
The treatments reduced the risk of hospitalization by 70 percent when given within 10 days of the onset of symptoms, Dr. Michael Anderson, a senior adviser to the U.S. Department of Health and Human Services, said during a briefing to Nevada news media in August.
Monoclonal antibodies are molecules produced in laboratories that act as substitute antibodies, mimicking the immune system’s response to infection. They are created by exposing a white blood cell to a particular viral protein, and then cloning the cell to produce antibodies that target the virus.
Partnering with the federal government, Sunrise hospital opened the third federally assisted center in the U.S. last January, during the most devastating wave of COVID-19 to date.
Since then, Sunrise workers have treated 1,850 patients with the therapy and “truly believe that it did keep our patients out of the hospital,” Merta said. However, he had no hospital-specific data.
Monday’s announcement from the FDA was expected, as both Regeneron and Eli Lilly have said for weeks that their treatments are less able to target omicron because of its mutations. The FDA, which pulled its emergency-use authorization for the drugs, said omicron is responsible for more than 99 percent of new infections in the U.S., making it “highly unlikely” that the antibodies would be effective for people seeking these treatments.
In updated drug labeling released Monday, the FDA said omicron appears 1,000-fold less vulnerable to Regeneron’s drug and nearly 3,000-fold less vulnerable to Eli Lilly’s drug.
As at Sunrise, the Outpatient Monoclonal Antibody Clinic at University Medical Center stopped administering the Regeneron and Eli Lilly therapies “several weeks ago,” representative Scott Kerbs said.
The Las Vegas hospital is focusing instead on providing high-risk patients with sotrovimab as well as Pfizer’s Paxlovid antiviral pills, he said.
“While supplies of sotrovimab and Paxlovid are limited, UMC remains committed to providing high-risk patients with access to latest breakthroughs in COVID-19 treatment,” Kerbs said in an email.
Nevada’s allocation from the federal government of sotrovimab is “very limited,” said a representative of the state Department of Health and Human Services. Nevada, which is receiving weekly allocations, got 360 doses last week, according to representative Shannon Litz.
The state has allocated tens of millions of federal dollars to provide monoclonal antibody treatments to Nevadans.
On Dec. 21, the Legislature’s Interim Finance Committee approved using $19.6 million in pandemic relief funds to set up new standalone monoclonal antibody treatment centers. In October the state Board of Examiners approved a federally funded $30 million contract for mobile treatments in rural areas.
As therapeutics are shown to be effective, “they will be incorporated into the State’s strategy of providing treatment options and information to the public,” Litz wrote in an email. “The State of Nevada is supporting all therapeutic treatments that are shown to be effective against the current variant of COVID-19. At this time only Sotrovimab monoclonal antibody treatment will be supported at the state-funded sites.”
Other treatments for omicron
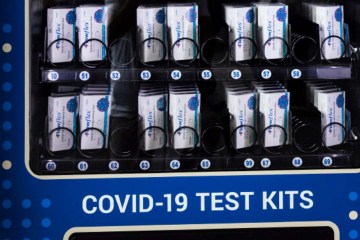
On Wednesday, the Southern Nevada Health District said it is expanding eligibility for its limited supply of the Paxlovid antiviral pills to a wider age range of patients who test positive at its Decatur Boulevard clinic.
The district is now offering the pills, which can be taken at home to reduce the risk of serious illness, to positive patients ages 12 to 64 who are symptomatic and have underlying health conditions. Patients 65 and older who test positive and are experiencing symptoms remain eligible for the pill.
Treatment with the drug cocktail of two different medications consists of taking 30 tablets over five days. The FDA does not recommend Paxlovid for patients with severe kidney or liver impairment.
The health district currently has 100 treatment courses of Paxlovid, Chief Medical Officer Dr. Cort Lohff said during a briefing.
Meanwhile, Florida Gov. Ron DeSantis on Tuesday pushed for coronavirus patients to continue receiving the Regeneron and Eli Lilly antibody treatments, vowing to fight White House health regulators in his latest feud with President Joe Biden.
“People have a right to access these treatments, and to revoke it on this basis is just fundamentally wrong, and we’re going to fight back,” DeSantis said at a news conference.
Contact Mary Hynes at mhynes@reviewjournal.com or 702-383-0336. Follow @MaryHynes1 on Twitter. The Associated Press contributed to this report.